FOR US HEALTHCARE PROFESSIONALS ONLY
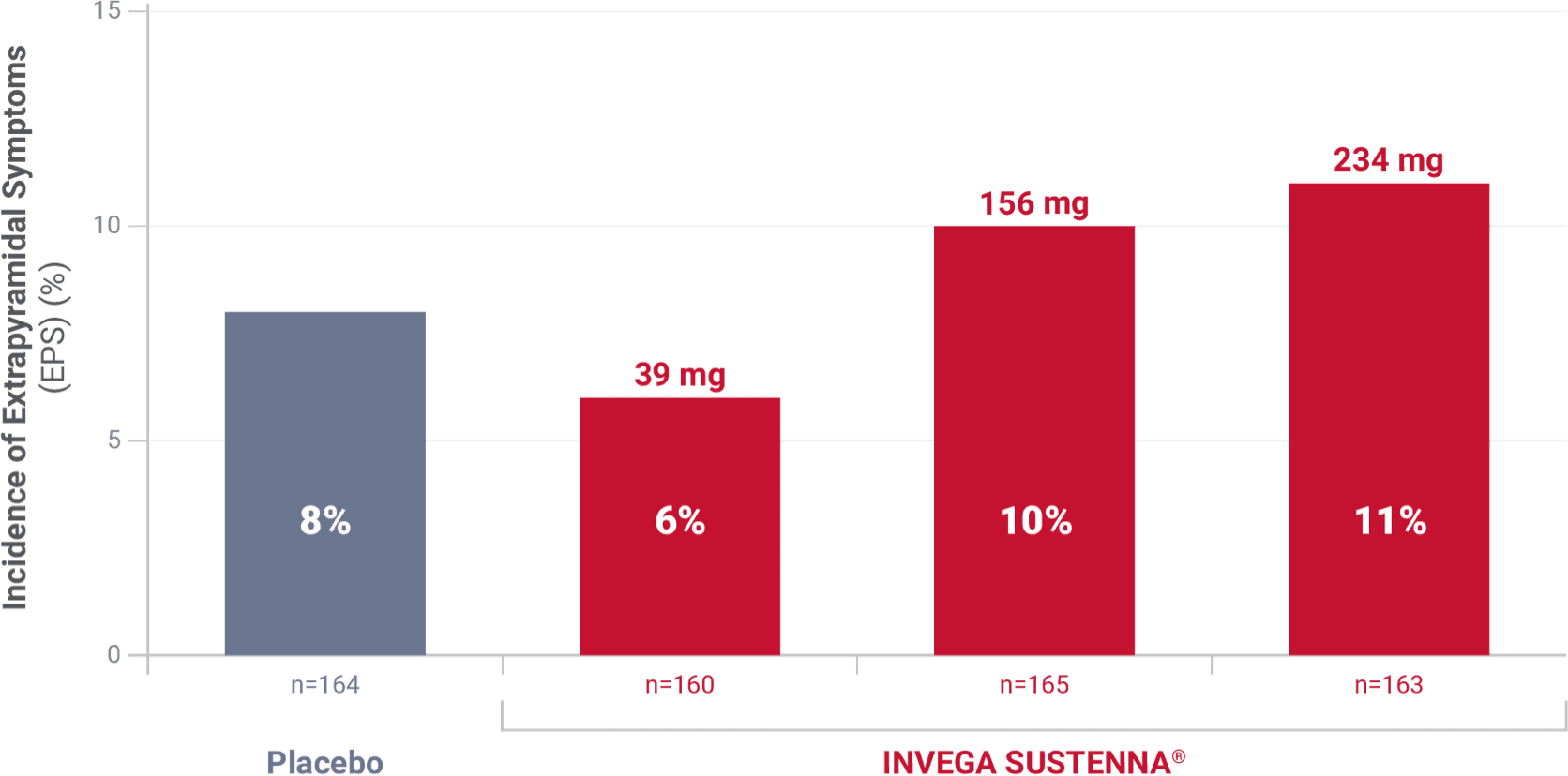
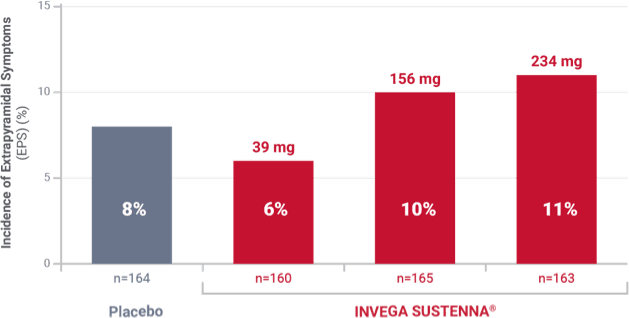
Incidence of extrapyramidal symptoms in a 13-week study of adult patients experiencing an acute exacerbation of schizophrenia1
Short-term (13-week) study design
Results from a double-blind, randomized, placebo-controlled, fixed-dose, 13-week study of adult inpatients experiencing an acute exacerbation of schizophrenia. Patients were randomized to receive placebo or a 234-mg deltoid injection initiation dose on Day 1, followed by a 39-mg, 156-mg, or 234-mg dose in either the deltoid or gluteal muscle on Day 8, and once monthly thereafter.1,2
Additional short-term study outcomes
- Pooled data from two 13-week, double-blind studies showed that the overall percentages of EPS-related adverse events were 10% in the placebo group and 12%, 11%, and 11% in the INVEGA SUSTENNA® 39-mg, 78-mg, and 156-mg groups, respectively1
- In a 9-week, double-blind study, the incidence of parkinsonism and akathisia was higher in the INVEGA SUSTENNA® 156-mg group (18% and 11%, respectively) than in the INVEGA SUSTENNA® 78-mg group (9% and 5%, respectively) and placebo group (7% and 4%, respectively)1
- The results across all phases of the maintenance trial exhibited comparable findings1
References: 1. INVEGA SUSTENNA® [Prescribing Information]. Titusville, NJ: Janssen Pharmaceuticals, Inc. 2. Pandina GJ, Lindenmayer J-P, Lull J, et al. A randomized, placebo-controlled study to assess the efficacy and safety of 3 doses of paliperidone palmitate in adults with acutely exacerbated schizophrenia. J Clin Psychopharmacol. 2010;30(3):235-244.
Back to Top