FOR US HEALTHCARE PROFESSIONALS ONLY
INVEGA SUSTENNA® is the ONLY* LAI to have demonstrated statistical superiority vs a group of 7 commonly prescribed oral antipsychotics1,2†
In a 15-month study with a real-world design that included patients typically excluded from clinical trials and was representative of patients you may see in your practice, INVEGA SUSTENNA® demonstrated1,2†:
*Only Johnson & Johnson conducted this clinical trials.
Primary endpoint
30%
lower risk of first treatment failure1‡
>6-month-longer delay
in time to relapse1द
Secondary endpoint
~15 months
(>450 days)
median time to first psychiatric hospitalization or arrest/incarceration was not reached2
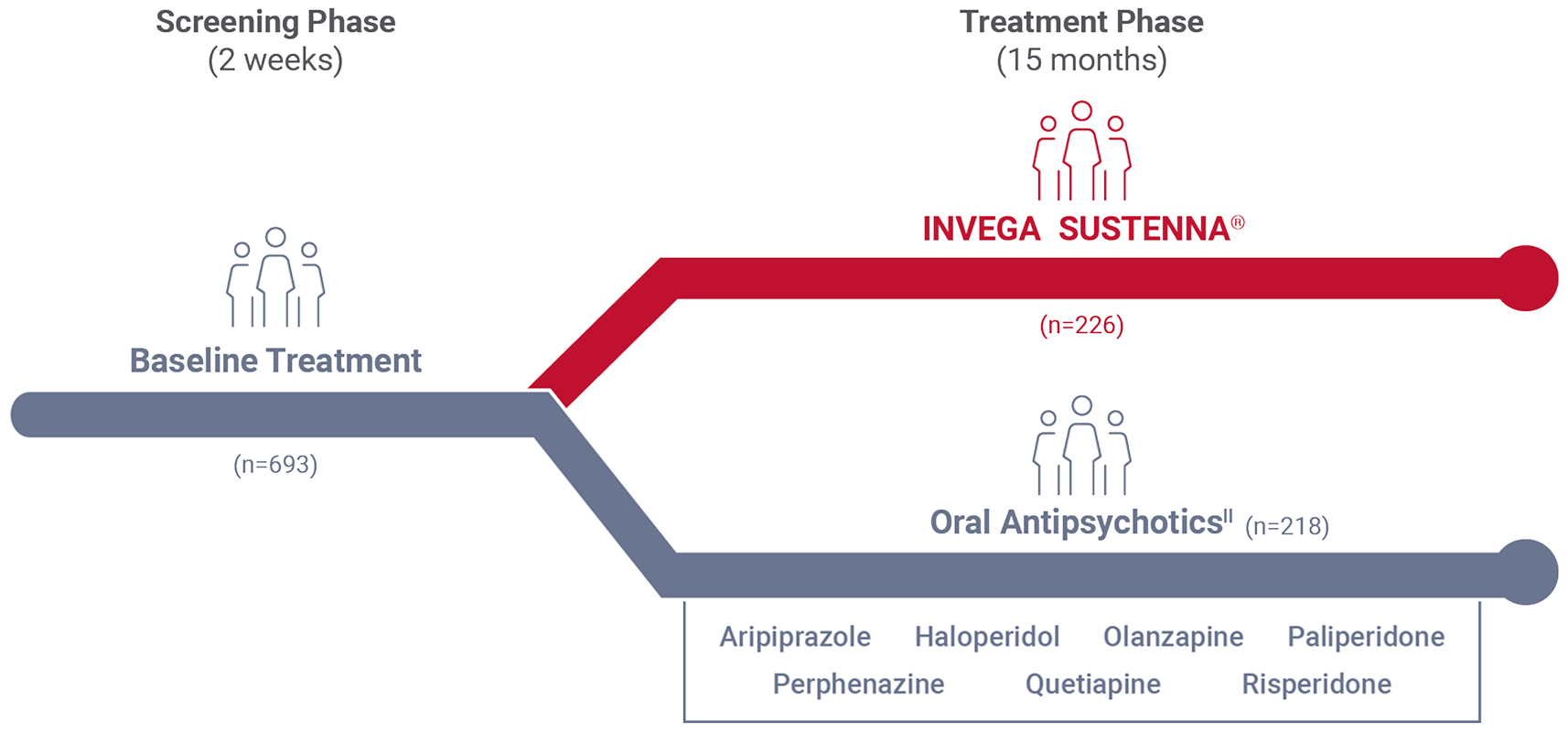
The 7 commonly prescribed orals in the study: aripiprazole, haloperidol, olanzapine, paliperidone, perphenazine, quetiapine, risperidone1
- The study was not powered to compare the efficacy of INVEGA SUSTENNA® with that of individual oral antipsychotics
- Data from randomization until end of randomly assigned treatment (28 days after last injection of INVEGA SUSTENNA® or 1 day after last dose of oral antipsychotic). Population included adults diagnosed with schizophrenia within 5 years of study entry randomized to 1 of 7 oral antipsychotics2
- Percentage of patients who discontinued due to adverse events was 11.9% in the INVEGA SUSTENNA® group and 7.8% in the oral antipsychotic group2
- The 5 most common adverse events in the INVEGA SUSTENNA® group were: injection-site pain (18.6%), insomnia (16.8%), weight increase (11.9%), akathisia (11.1%), and anxiety (10.6%)2
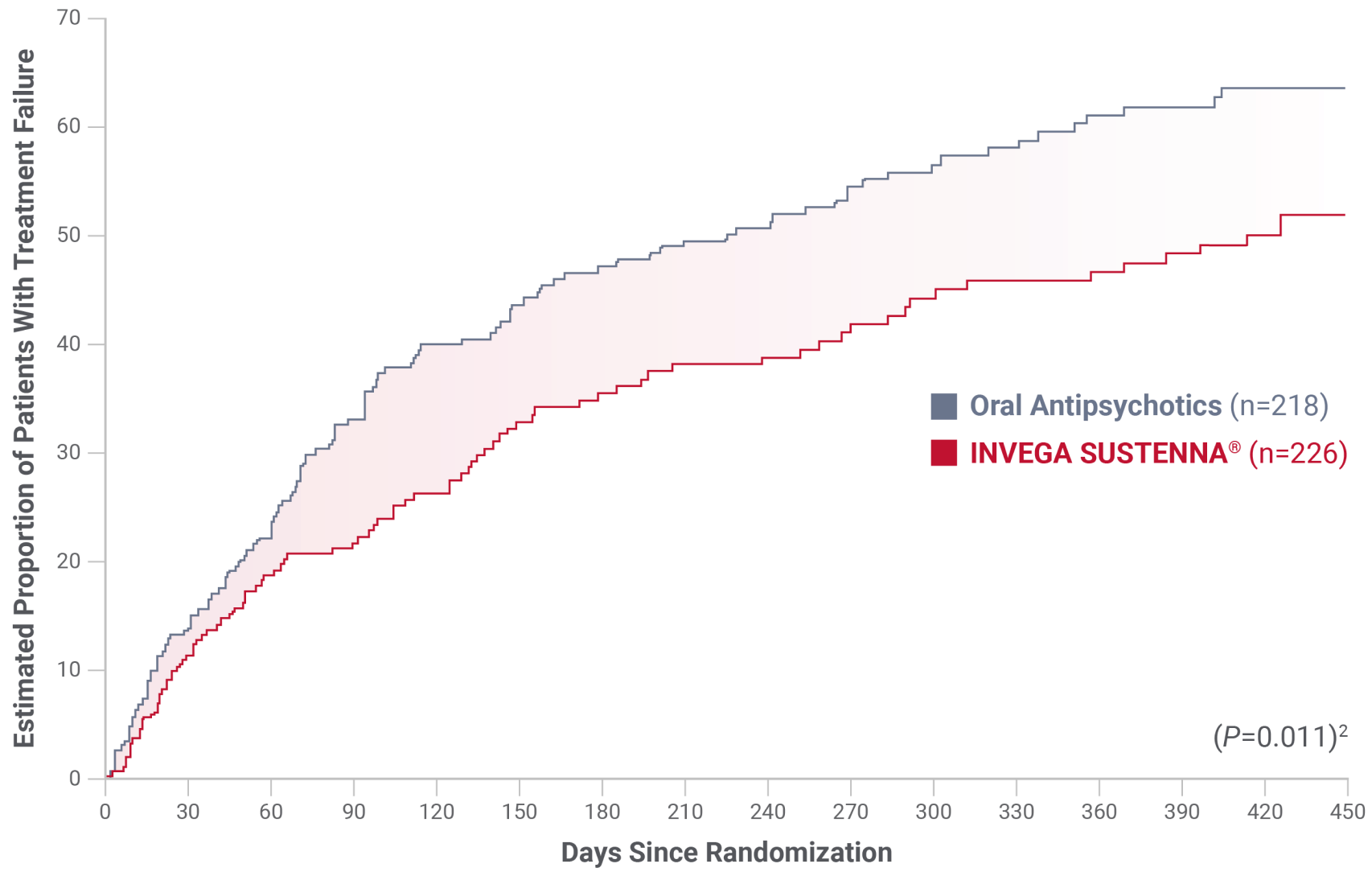
- Time to first treatment failure, defined as 1 of the following1: psychiatric hospitalization, arrest/incarceration, discontinuation of antipsychotic treatment because of safety or tolerability concerns, treatment supplementation with another antipsychotic because of inadequate efficacy, increase in level of psychiatric services to prevent an imminent psychiatric hospitalization, discontinuation of antipsychotic treatment because of inadequate efficacy, and suicide1
LAI=long-acting injectable.
†The 7 oral antipsychotics included in the comparative arm accounted for 74% of oral schizophrenia treatment during the study period.3
‡Real-world was defined by patient selection and clinically meaningful outcome measures.2
§Relapse was defined as time to first treatment failure.1
¶Median time to first treatment failure in the INVEGA SUSTENNA® group was 416 days vs 226 days in the oral antipsychotic group.1
INVEGA SUSTENNA® demonstrated a 6-month-longer delay in time to relapse vs a group of oral antipsychotics1
Primary endpoint: Time to first treatment failure in a long-term, randomized, flexible-dose study in adult patients with schizophrenia and a history of incarceration1
Select real-world* patient characteristics relevant to clinical practice† included adults living with schizophrenia often excluded from trials to compare how antipsychotics work in clinical practice2
59.5%
Patients with comorbid substance abuse2
42.2 days
Mean time since release from last incarceration2
- The study was not powered to compare the efficacy of INVEGA SUSTENNA® with that of individual oral antipsychotics
- Substance abuse was not exclusionary, but subjects who had abused intravenous drugs within 3 months of screening or had an opiate dependence disorder (DSM-IV®) were excluded2
- Percentage of patients who discontinued due to adverse events was 11.9% in the INVEGA SUSTENNA® group and 7.8% in the oral antipsychotic group2
- The 5 most common adverse events in the INVEGA SUSTENNA® group were: injection-site pain (18.6%), insomnia (16.8%), weight increase (11.9%), akathisia (11.1%), and anxiety (10.6%)2
Relapse criteria
Time to first treatment failure, defined as 1 of the following2:
- Psychiatric hospitalization
- Arrest/Incarceration
- Discontinuation of antipsychotic treatment because of safety or tolerability concerns
- Treatment supplementation with another antipsychotic because of inadequate efficacy
- Increase in level of psychiatric services to prevent an imminent psychiatric hospitalization
- Discontinuation of antipsychotic treatment because of inadequate efficacy
- Suicide
DSM-IV=Diagnostic and Statistical Manual of Mental Disorders, 4th ed.
*Real-world was defined by patient selection and clinically meaningful outcome measures.2
†The 7 oral antipsychotics included in the comparative arm accounted for 74% of oral schizophrenia treatment during the study period.3
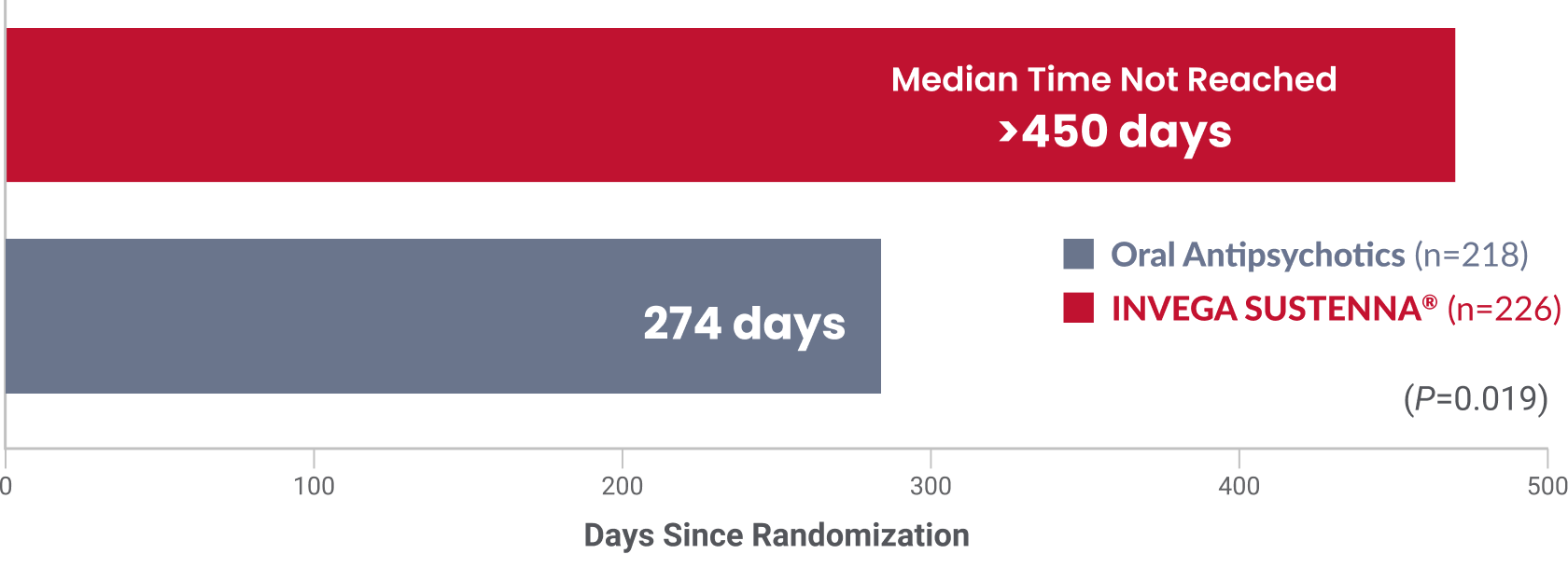
With INVEGA SUSTENNA®, the median time to first psychiatric hospitalization or arrest/incarceration was not reached at 15 months2
Secondary endpoint: Time to first psychiatric hospitalization or arrest/incarceration was significantly longer vs commonly prescribed orals2
- The study was not powered to compare the efficacy of INVEGA SUSTENNA® with that of individual oral antipsychotics
- Median time to first psychiatric hospitalization or arrest/incarceration was not reached in the INVEGA SUSTENNA® group (>450 days)2
- Median time to first psychiatric hospitalization or arrest/incarceration in the oral antipsychotic group was 274 days2
Study notes
Adult patients assigned to the INVEGA SUSTENNA® group were initiated with 2 injections in the deltoid muscle that were given approximately 1 week apart: 234 mg on Day 1 and 156 mg on Day 8 (±4 days)2
- Flexible monthly maintenance dosing for INVEGA SUSTENNA® was started on Day 38 within a range of 78 mg to 234 mg (median dose was 156 mg)1,2
- Before randomization, each patient and the treating clinician could deselect up to 6 of the 7 oral antipsychotics based on prior patient experience2
- Patients not previously treated with paliperidone palmitate, paliperidone, or risperidone underwent 2-day oral tolerability testing with risperidone, 1 mg QD3
- Oral antipsychotic doses were given and adjusted within the range of the package insert2*
- 17% of adults were diagnosed within 5 years (the mean age of all patients in the study was 38 years [n=444])2,3
QD=every day.
*Occasional dosing outside of the package insert range was allowed.
Frequencies of first treatment failure events by type
Components of composite endpoint in a long-term, randomized, flexible-dose study in subjects with schizophrenia and a history of incarceration1
| Event type | INVEGA SUSTENNA® (n=226) frequency (%) | Oral Antipsychotics (n=218) frequency (%) | Hazard Ratio* [95% Cl] |
|---|---|---|---|
| First treatment failure | 90 (39.8%) | 117 (53.7%) | 0.70 [0.53, 0.92] |
| First treatment failure component events | |||
| Arrest and/or incarceration | 48 (21.2%) | 64 (29.4%) | |
| Psychiatric hospitalization | 18 (8.0%) | 26 (11.9%) | |
| Discontinuation of antipsychotic treatment because of safety or tolerability | 15 (6.6%) | 8 (3.7%) | |
| Treatment supplementation with another antipsychotic because of inadequate efficacy | 5 (2.2%) | 6 (2.8%) | |
| Need for increase in level of psychiatric services to prevent imminent psychiatric hospitalization | 3 (1.3%) | 4 (1.8%) | |
| Discontinuation of antipsychotic treatment because of inadequate efficacy | 1 (0.4%) | 9 (4.1%) | |
| Suicide | 0 | 0 | |
| Arrest and/or incarceration or psychiatric hospitalization events, regardless of whether they were first events† | 76 (33.6%) | 98 (45.0%) | 0.70 [0.52, 0.94] |
CI=confidence interval.
*Hazard ratio of INVEGA SUSTENNA® to oral antipsychotics based on Cox regression model for time-to-event analysis. Note that the hazard ratio did not appear constant throughout the trial.
†Analysis results, which incorporated relevant events collected after discontinuation for those who discontinued, were consistent with the results from the prespecified analysis of this secondary endpoint.
Real-world* study design included patients typically excluded from clinical trials and was representative of patients you may see in your practice2
Comorbid substance abuse†
~60%
of patients had comorbid
substance abuse
History of incarceration‡
100%
of patients had been incarcerated and released within 90 days of study onset
Unstable living conditions§
~14%
of patients
were homeless
Additional factors that contributed to making this study a real-world reflection of how treatments are managed in practice2
- Study medications could be flexibly dosed within the range indicated in the Prescribing Information for each product
- Before randomization, HCPs and patients worked together to review treatment options and were permitted to exclude up to 6 oral medications based on patient history
- Oral medication prescribing, distribution, and adherence were monitored, but patient adherence was not mandated
- Medication adherence was monitored but not required, resulting in study conditions that better reflected real-world clinical practice
Comparative efficacy study design

*Real-world was defined by patient selection and clinically meaningful outcome measures.2
†Except for patients who had abused intravenous drugs within 3 months of screening or had an opiate-dependence disorder (DSM-IV®).2
‡Patients must have been arrested ≥2 times in the previous 2 years, with ≥1 event leading to incarceration; released from most recent custody within 90 days of the screening visit.2
§Homelessness was defined as living on the streets or in an emergency shelter for the homeless since the time of release from jail.2
||Data from randomization until end of randomly assigned treatment (28 days after last injection of INVEGA SUSTENNA® or 1 day after last dose of oral antipsychotic). Population included adults diagnosed with schizophrenia within 5 years of study entry randomized to 1 of 7 oral antipsychotics.2 The 7 oral antipsychotics included in the comparative arm accounted for 74% of oral schizophrenia treatment during the study period.3
Key inclusion criteria1,2
- Adults (18 to 65 years old)
- Current diagnosis of schizophrenia (DSM-IV® criteria)
- Patients must have been taken into criminal justice system custody ≥2 times in the previous 2 years, with ≥1 of these events leading to incarceration
- Released from most recent custody within 90 days of screening
Key exclusion criteria2
- Use of clozapine within 3 months of screening or an injectable antipsychotic within 2 injection cycles of screening
- Substance abuse was not exclusionary, but subjects who had abused intravenous drugs within 3 months of screening or had an opiate dependence disorder (DSM-IV®) were excluded
Primary endpoint1
Median time to first treatment failure in the INVEGA SUSTENNA® group was 416 days vs 226 days in the oral antipsychotic group.
Secondary endpoint2
Median time to first psychiatric hospitalization or arrest/incarceration was not reached in the INVEGA SUSTENNA® group (>450 days). Median time to first psychiatric hospitalization or arrest/incarceration in the oral antipsychotic group was 274 days.2
Real-world design1,2
Real-world design elements
- Included patients who are typically excluded from clinical trials:
- Comorbid substance abuse*
- History of incarceration†
- Unstable living conditions
- Flexible treatment interventions
- OraI antipsychotics could be deselected prior to randomization
- Allowed dosing flexibility and use of concomitant medications
- Medication adherence was monitored but not required to complete the trial
Clinical trial features
- Prospective
- Randomized
- Open-label with blinded Event Monitoring Board
- A 15-month, head-to-head trial vs a group of 7 commonly prescribed oral antipsychotics‡
DSM-IV=Diagnostic and Statistical Manual of Mental Disorders, 4th ed.
*Except for patients who had abused intravenous drugs within 3 months of screening or had an opiate-dependence disorder (DSM-IV®).2
†Patients must have been arrested ≥2 times in the previous 2 years, with ≥1 event leading to incarceration; released from most recent custody within 90 days of the screening visit.2
‡The 7 oral antipsychotics included in the comparative arm accounted for 74% of oral schizophrenia treatment during the study period.3
Back to Top