INDICATION
INVEGA SUSTENNA® (paliperidone palmitate) is indicated for the treatment of:
- Schizoaffective disorder as monotherapy and as an adjunct to mood stabilizers or antidepressants in adults.
IMPORTANT SAFETY INFORMATION FOR INVEGA SUSTENNA® (paliperidone palmitate)
WARNING: INCREASED MORTALITY IN ELDERLY PATIENTS WITH DEMENTIA-RELATED PSYCHOSIS.
See full prescribing information for complete Boxed Warning.
Elderly patients with dementia-related psychosis treated with antipsychotic drugs are at an increased risk of death. INVEGA SUSTENNA® is not approved for use in patients with dementia-related psychosis.
Contraindications: INVEGA SUSTENNA® is contraindicated in patients with a known hypersensitivity to either paliperidone, risperidone, or to any excipients of the INVEGA SUSTENNA® formulation.
Cerebrovascular Adverse Reactions: Cerebrovascular adverse reactions (e.g., stroke, transient ischemic attacks), including fatalities, were reported at a higher incidence in elderly patients with dementia-related psychosis taking risperidone, aripiprazole, and olanzapine compared to placebo. No studies have been conducted with oral paliperidone, INVEGA SUSTENNA®, or the 3-month paliperidone palmitate extended-release injectable suspension in elderly patients with dementia. These medicines are not approved for the treatment of patients with dementia-related psychosis.
Neuroleptic Malignant Syndrome (NMS): NMS, a potentially fatal symptom complex, has been reported in association with antipsychotic drugs, including paliperidone.
Clinical manifestations of NMS are hyperpyrexia, muscle rigidity, altered mental status including delirium, and autonomic instability (irregular pulse or blood pressure, tachycardia, diaphoresis, and cardiac dysrhythmia). Additional signs may include elevated creatine phosphokinase, myoglobinuria (rhabdomyolysis), and acute renal failure.
If NMS is suspected, immediately discontinue INVEGA SUSTENNA® and provide symptomatic treatment and monitoring.
QT Prolongation: Paliperidone causes a modest increase in the corrected QT (QTc) interval. Avoid the use of drugs that also increase QTc interval and in patients with risk factors for prolonged QTc interval. Paliperidone should also be avoided in patients with congenital long QT syndrome and in patients with a history of cardiac arrhythmias. Certain circumstances may increase the risk of the occurrence of torsades de pointes and/or sudden death in association with the use of drugs that prolong the QTc interval.
Tardive Dyskinesia (TD): TD, a syndrome consisting of potentially irreversible, involuntary, dyskinetic movements, may develop in patients treated with antipsychotic drugs. Although the prevalence of the syndrome appears to be highest among the elderly, especially elderly women, it is impossible to predict which patients will develop the syndrome. Whether antipsychotic drug products differ in their potential to cause tardive dyskinesia is unknown.
The risk of developing TD and the likelihood that it will become irreversible appear to increase with the duration of treatment and the cumulative dose. The syndrome can develop after relatively brief treatment periods, even at low doses. It may also occur after discontinuation. TD may remit, partially or completely, if antipsychotic treatment is discontinued. Antipsychotic treatment itself, however, may suppress (or partially suppress) the signs and symptoms of the syndrome, possibly masking the underlying process. The effect that symptomatic suppression has upon the long-term course of the syndrome is unknown.
If signs and symptoms of TD appear in a patient on INVEGA SUSTENNA®, drug discontinuation should be considered. However, some patients may require treatment with INVEGA SUSTENNA® despite the presence of the syndrome. In patients who do require chronic treatment, use the lowest dose and the shortest duration of treatment producing a satisfactory clinical response. Periodically reassess the need for continued treatment.
Metabolic Changes: Atypical antipsychotic drugs have been associated with metabolic changes that may increase cardiovascular/cerebrovascular risk. These metabolic changes include hyperglycemia, dyslipidemia, and body weight gain. While all of the drugs in the class have been shown to produce some metabolic changes, each drug has its own specific risk profile.
Hyperglycemia and Diabetes Mellitus: Hyperglycemia and diabetes mellitus, in some cases extreme and associated with ketoacidosis, hyperosmolar coma or death, have been reported in patients treated with all atypical antipsychotics (APS). Patients starting treatment with APS who have or are at risk for diabetes mellitus should undergo fasting blood glucose testing at the beginning of and during treatment. Patients who develop symptoms of hyperglycemia during treatment should also undergo fasting blood glucose testing. All patients treated with atypical antipsychotics should be monitored for symptoms of hyperglycemia. Some patients require continuation of antidiabetic treatment despite discontinuation of the suspect drug.
Dyslipidemia: Undesirable alterations have been observed in patients treated with atypical antipsychotics.
Weight Gain: Weight gain has been observed with atypical antipsychotic use. Clinical monitoring of weight is recommended.
Orthostatic Hypotension and Syncope: INVEGA SUSTENNA® may induce orthostatic hypotension in some patients due to its alpha-adrenergic blocking activity. INVEGA SUSTENNA® should be used with caution in patients with known cardiovascular disease, cerebrovascular disease or conditions that would predispose patients to hypotension (e.g., dehydration, hypovolemia, treatment with antihypertensive medications). Monitoring should be considered in patients for whom this may be of concern.
Falls: Somnolence, postural hypotension, motor and sensory instability have been reported with the use of antipsychotics, including INVEGA SUSTENNA®, which may lead to falls and, consequently, fractures or other fall-related injuries. For patients, particularly the elderly, with diseases, conditions, or medications that could exacerbate these effects, assess the risk of falls when initiating antipsychotic treatment and recurrently for patients on long-term antipsychotic therapy.
Leukopenia, Neutropenia and Agranulocytosis have been reported with antipsychotics, including INVEGA SUSTENNA®. In patients with a history of clinically significant low white blood cell count (WBC)/absolute neutrophil count (ANC) or drug-induced leukopenia/neutropenia, perform a complete blood count frequently during the first few months of therapy. Consider discontinuing INVEGA SUSTENNA® at the first sign of a clinically significant decline in WBC in the absence of other causative factors. Monitor patients with clinically significant neutropenia for fever or other symptoms or signs of infection and treat promptly if such symptoms or signs occur. Discontinue INVEGA SUSTENNA® in patients with severe neutropenia (absolute neutrophil count <1000/mm3) and follow their WBC until recovery.
Hyperprolactinemia: As with other drugs that antagonize dopamine D2 receptors, INVEGA SUSTENNA® elevates prolactin levels, and the elevation persists during chronic administration. Paliperidone has a prolactin-elevating effect similar to risperidone, which is associated with higher levels of prolactin elevation than other antipsychotic agents.
Potential for Cognitive and Motor Impairment: Somnolence, sedation, and dizziness were reported as adverse reactions in subjects treated with INVEGA SUSTENNA®.
INVEGA SUSTENNA® has the potential to impair judgment, thinking, or motor skills. Patients should be cautioned about performing activities that require mental alertness such as operating hazardous machinery, including motor vehicles, until they are reasonably certain that INVEGA SUSTENNA® does not adversely affect them.
Seizures: INVEGA SUSTENNA® should be used cautiously in patients with a history of seizures or with conditions that potentially lower seizure threshold. Conditions that lower seizure threshold may be more prevalent in patients 65 years or older.
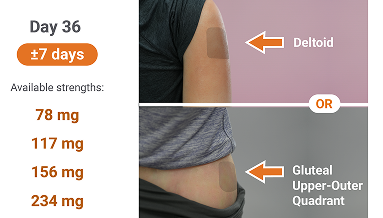
Administration: For intramuscular injection only by a healthcare professional using only the needles provided in the INVEGA SUSTENNA® kit. Care should be taken to avoid inadvertent injection into a blood vessel.
Drug Interactions: Strong CYP3A4/P-glycoprotein (P-gp) inducers: Avoid using a strong inducer of CYP3A4 and/or P-gp (e.g. carbamazepine, rifampin, St. John’s Wort) during a dosing interval for INVEGA SUSTENNA®. If administering a strong inducer is necessary, consider managing the patient using paliperidone extended-release tablets.
Pregnancy/Nursing: INVEGA SUSTENNA® may cause extrapyramidal and/or withdrawal symptoms in neonates with third trimester exposure. Advise patients to notify their healthcare professional if they become pregnant or intend to become pregnant during treatment with INVEGA SUSTENNA®. Patients should be advised that there is a pregnancy registry that monitors outcomes in women exposed to INVEGA SUSTENNA® during pregnancy. INVEGA SUSTENNA® can pass into human breast milk. The benefits of breastfeeding should be considered along with the mother’s clinical need for INVEGA SUSTENNA® and any potential adverse effects on the breastfed infant from INVEGA SUSTENNA® or the mother’s underlying condition.
Commonly Observed Adverse Reactions for INVEGA SUSTENNA®: No adverse events occurred at a rate of ≥5% and twice placebo during the 15 month double-blind, placebo-controlled study in patients with schizoaffective disorder. The following adverse reactions occurred more frequently (a ≥2% difference vs. placebo) in the long-term study in patients with schizoaffective disorder: weight increased, nasopharyngitis, headache, hyperprolactinemia, and pyrexia.
Please click hereto read the full Prescribing Information, including Boxed WARNING, for INVEGA SUSTENNA®.
cp-64201v3
![Dosing chart showing initiation and monthly maintenance doses with injection sites on the body.]](/_next/static/media/dosing-2.0c6082f2.png)