Click here to start your patients
on the path to just 2 DOSES A YEAR
- For US Healthcare Professionals Only
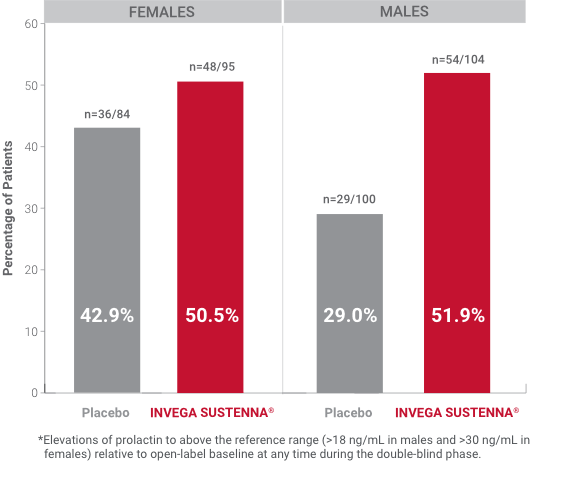
Hyperprolactinemia: As with other drugs that antagonize dopamine D2 receptors, INVEGA SUSTENNA® elevates prolactin levels, and the elevation persists during chronic administration. Paliperidone has a prolactin-elevating effect similar to risperidone, which is associated with higher levels of prolactin elevation than other antipsychotic agents.1
Please see Important Safety Information.
*Elevations of prolactin to above the reference range (>18 ng/mL in males and >30 ng/mL in females) relative to open-label baseline at any time during the double-blind phase.
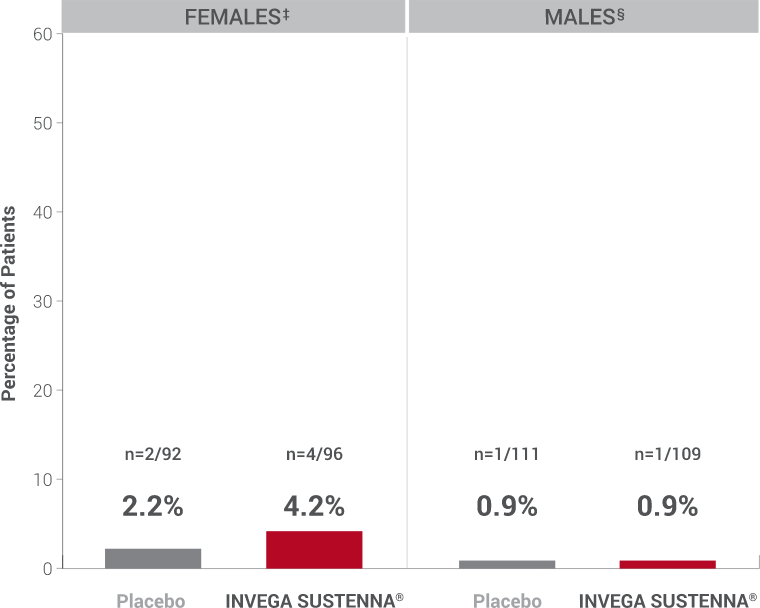
Hyperprolactinemia: As with other drugs that antagonize dopamine D2 receptors, INVEGA SUSTENNA® elevates prolactin levels, and the elevation persists during chronic administration. Paliperidone has a prolactin-elevating effect similar to risperidone, which is associated with higher levels of prolactin elevation than other antipsychotic agents.1
Please see Important Safety Information.
†Prolactin adverse reaction reports among all patients in the double-blind phase, regardless of baseline prolactin levels.
‡In the INVEGA SUSTENNA® group, 2 females reported amenorrhea, 1 reported galactorrhea, and 1 reported irregular menstruation, while in the placebo group, 1 female reported amenorrhea and 1 reported breast pain.
§In the INVEGA SUSTENNA® group, 1 male reported erectile dysfunction, and in the placebo group, 1 male reported gynecomastia.
Reference: 1. INVEGA SUSTENNA® [Prescribing Information]. Titusville, NJ: Janssen Pharmaceuticals, Inc.; July 2022.